Which is Better for Analytical Testing: pH Meter or pH Test Strip?

Compare pH meters and test strips to understand their accuracy, precision, strengths, and ideal applications for reliable analytical testing in labs.
When measuring the pH of a solution, laboratories generally rely on two tools: pH test strips, also known as pH indicator strips, and pH meters. Both can determine the hydrogen ion concentration of a liquid sample and are used in laboratories and fieldwork. However, they have their own strengths and limitations and differ greatly in terms of precision, performance, and ideal applications. This explains why knowing which tool to use is important, especially in analytical testing where accuracy and precision are vital.
pH Test Strips
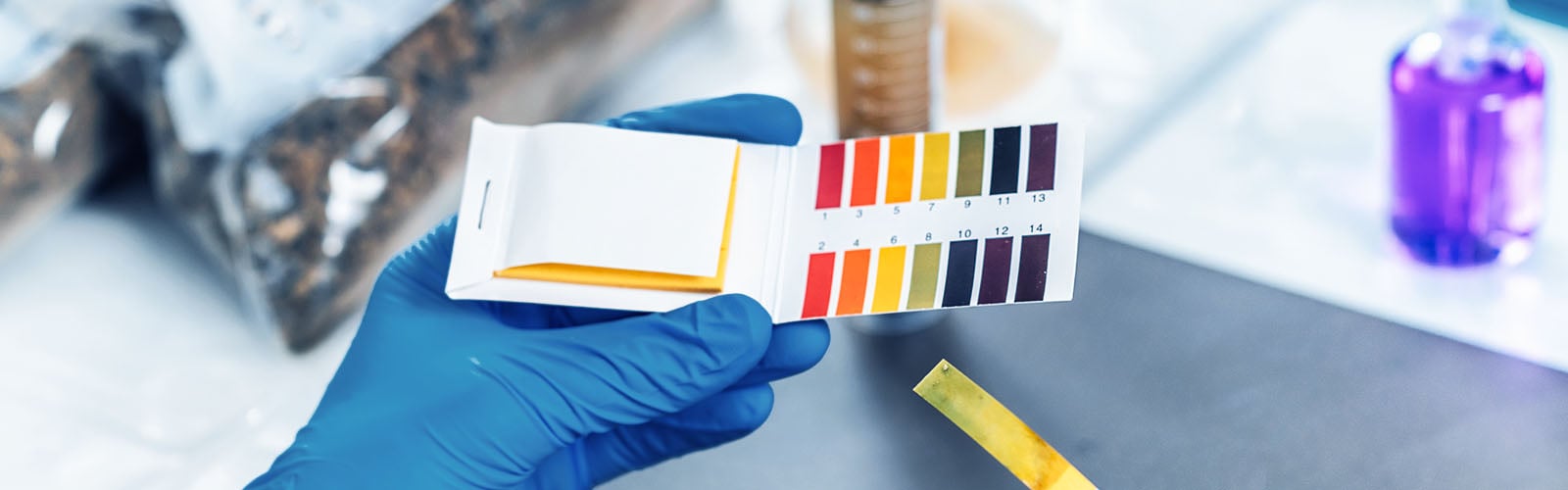
pH test strips are single-use indicators that contain a chemical dye. When they come in contact with a liquid sample, they almost instantly change color. Based on the reference chart, the color then provides an approximation of how acidic or basic the sample is.
The rapid results make these test strips a more convenient method for pH measurement. Add to this their other beneficial features, such as portability, disposability, and the need for no calibration, which is essential for pH meters. These test strips also cost less and do not require technical expertise, so much so that they are normally found in academic labs, manufacturing lines, and basic water tests.
However, the lack of precision is where pH test strips falter. They typically measure only within ±0.5 to ±1.0 pH units, which is considerably low in analytical testing. Interpretation of the results is also subjective and prone to human error, even under ideal lighting. Furthermore, results can be affected by the color and viscosity of the sample. In addition, they do not offer traceability.
Given their lower level of accuracy and precision, pH test strips are avoided in analytical testing. Instead, they can be used for preliminary checks and screening non-critical samples.
pH Meters
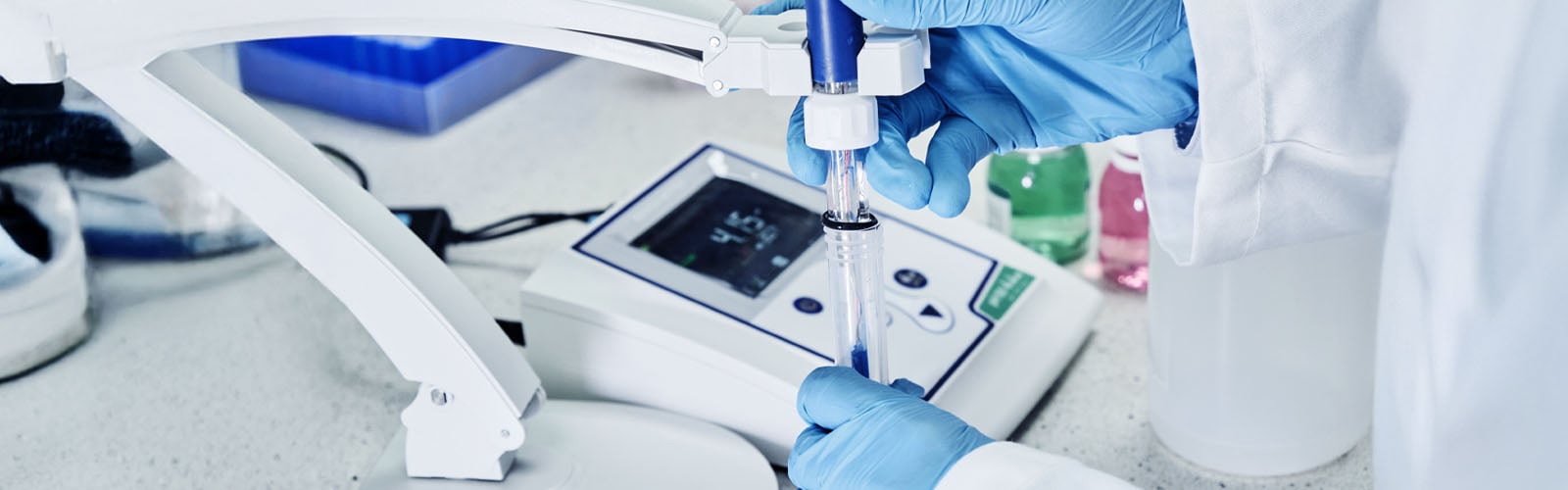
pH meters provide a more accurate measurement, which explains why they are the better choice in analytical and research labs. To determine a sample's acidity or alkalinity, pH meters use a glass electrode and a reference electrode to measure voltage differences, with a precision of ±0.01 pH units.
In addition to their ability to deliver accurate readings, benchtop and portable pH meters also offer automatic temperature compensation and calibration reminders, among other features commonly found in modern models. These instruments can also digitally export data.
However, the main drawback of pH meters is the high cost. Compared to disposable pH test strips, pH meters from trusted industry names like Thermo Scientific and OHAUS represent a higher upfront cost. On top of the upfront costs, they must be regularly calibrated and maintained. Calibrating them using standardized buffer solutions ensures their readings are accurate, while keeping the electrodes clean and hydrated keeps their ion-selective function effective.
Which Should You Use in Analytical Testing
| pH Test Strips | pH Meters | |
| Precision | ±0.5 to ±1.0 | ±0.01 |
| Ease of Use | Very simple | Moderate; training needed |
| Cost per Use | Low | Lower over time |
| Maintenance | None | Regular calibration |
| Traceability | None | Full digital documentation |
| Data Logging | None | Available in most digital pH meters |
| Best For | Quick field checks | Analytical and research applications |
| Regulatory Compliance | Limited | Meets QA/QC standards |
The comparison chart clearly shows that pH meters are ideal for analytical testing. Although test strips are more convenient, faster, and easier to use, they fail to provide the necessary precision, data integrity, and repeatability.
FAQs
What are the three types of pH meters?
The three main types are benchtop pH meters, which offer high precision and advanced features for laboratory use; portable pH meters, which provide reliable results for field or flexible lab testing; and pocket pH testers, which are compact devices ideal for quick, basic pH measurements.
How do I calibrate a pH meter?
For exact calibration or two- or three-point calibration, check the user manual of your pH meter. For one-point calibration, follow these steps:
- Turn on the meter, and allow the pH meter to stabilize before starting calibration
- Pour a small amount of fresh pH 7.0 buffer into a clean container. Never dip the electrode into the bottle.
- Use distilled or deionized water to rinse the electrode. Do not touch the glass bulb.
- Place the electrode in the buffer until the sensor tip is fully submerged. Allow the reading to stabilize.
- Press the Calibrate button and set the meter to match the buffer's pH 7.0 value. Wait for the meter to confirm calibration.
- Rinse the electrode again before measuring samples or returning it to the storage solution.
- Record the date of calibration, buffer used, and any observations.
Where do I buy pH test strips?
You can purchase them from reliable laboratory supply distributors, such as The Lab Depot. These strips can be used in laboratory, educational, and industrial applications.
What are the best pH test strips?
pH test strips that deliver consistent, accurate readings within ±0.2 to ±0.5 pH units and have a wide range (0-14) are preferred. Choose non-bleeding test strips, such as the pH-Fix pH Indicator Strips from CTL Scientific Supply, to ensure color accuracy and easy comparison, even with strongly buffered or colored samples.
The Lab Depot offers various pH meters and pH test strips from reliable lab manufacturers. Browse through our product inventory to find the products you need. For expert assistance, feel free to contact our team at 1-800-733-2522, email, or through live chat on our website.
Sources:
https://aperainst.com/blog/why-choose-a-ph-meter-instead-of-a-ph-test-strip/
https://infitek.com/exploring-the-role-of-ph-meters-in-the-laboratory/
https://hudsonrobotics.com/ph-meter-principles-and-applications/
https://www.drawellanalytical.com/mastering-ph-measurement-a-guide-to-ph-meters-use-in-laboratory/
https://www.omega.com/en-us/resources/ph-meter
https://www.coleparmer.com/blog/how-to-calibrate-ph-meter/
https://www.drawellanalytical.com/what-are-the-types-of-ph-meters-a-guide-to-ph-meter-selection/