pH Measurement 101: Techniques and Tools for Accurate Results

pH measurement is often a critical early step in your workflow. Thermo Scientific offers an extensive line of pH meters, pH electrodes, pH buffers, and pH measurement accessories designed to work seamlessly together to make your complex pH measurements routine and reliable. Measure with confidence with products you trust, like Orion and ROSS electrodes.
This guide outlines the critical points about pH measurement and explains the benefits of using Thermo Scientific instruments to measure and test a solution's pH.
What is pH?

pH is a scale used to measure the acidity or alkalinity of a solution at a given temperature. This parameter is crucial across various industries and fields, as it significantly influences chemical reactions, biological processes, and the behavior of substances in different environments.
The pH scale ranges from 0 to 14 and is classified into three categories: neutral, acidic, and basic.
- A pH of 7 describes a neutral solution because the activities of hydrogen and hydroxide ions are equal.
- When the pH is below 7, the solution is described as acidic—the hydrogen ion activity increases and the pH value decreases.
- When the pH is above 7, the solution is described as basic (or alkaline)—the hydroxide ions' activity is greater than the hydrogen ions.
How is pH Measured?
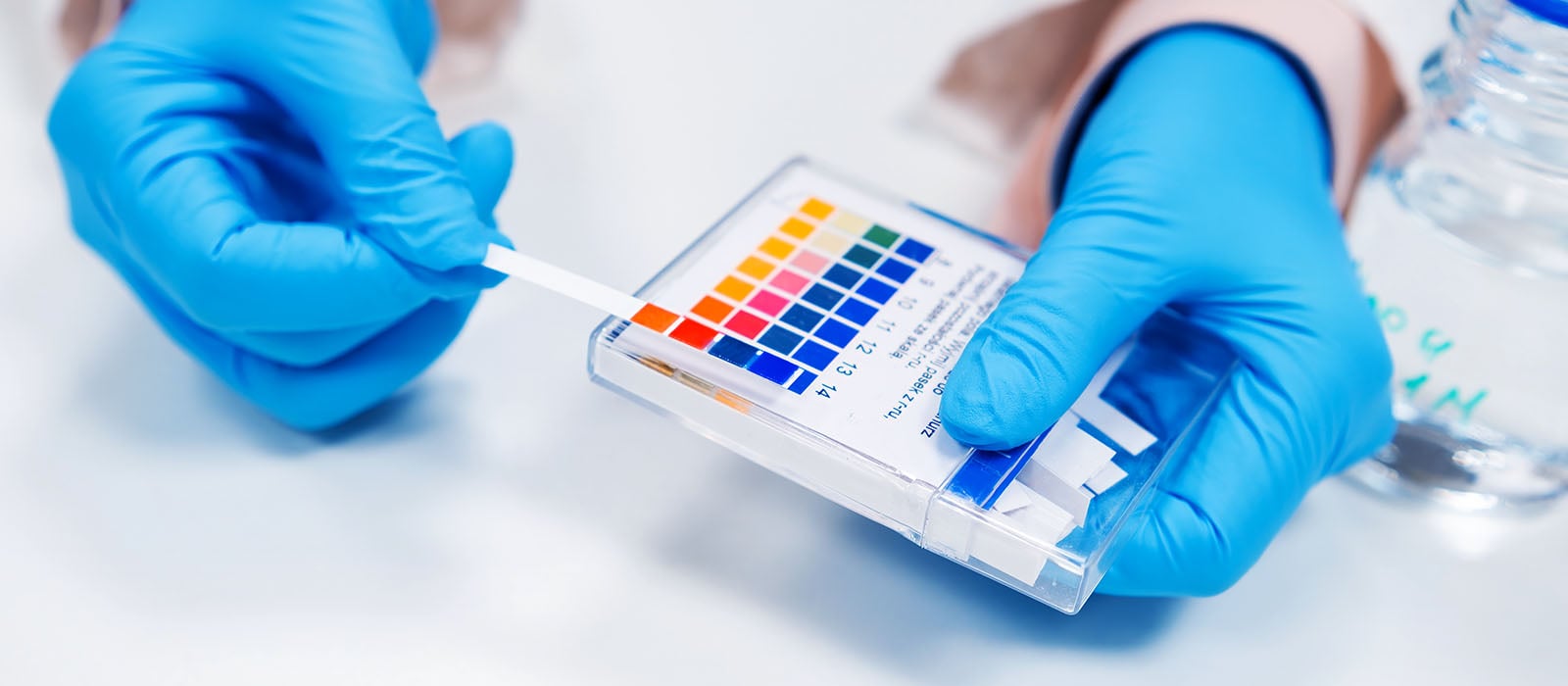
pH electrode measurements are obtained by comparing the readings in a sample with those in standards whose pH has been defined (buffers).
Thermo Scientific pH meters contain pH versus temperature values for commonly used buffers. This feature enables the meter to identify specific pH buffers and calibrate accurately with the correct buffer value at the measured temperature.
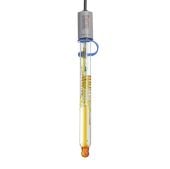
Anatomy of a pH Electrode

Explore the features of the Orion pH Electrode and understand how its advanced technology ensures precise and reliable pH measurements.
pH Measurement Products and Solutions

Thermo Scientific offers a range of pH measurement solutions to help you achieve successful lab processes with reproducible results:
- pH Meters - Dedicated pH testing in water, process, research, and QA/QC samples. Select from simple and economical models to powerful and sophisticated meters.
- pH Electrodes, Standards, and Solutions - Quality pH electrodes, standards, and solutions are available for various body materials, body styles, fill types, reference styles, and functions to meet your needs.
- pH and ORP Pocket Testers - Thermo Scientific Pocket Testers are ideal for on-the-go electrochemical testing.
- Online pH Water Measurement Systems - Thermo Scientific AquaSensors, Eutech, and Orion sensors provide real-time, accurate pH/ORP measurements and more across your facility.
What Causes an Error in pH Measurements?

The most common cause of error in pH measurements is temperature. Temperature variations can influence pH for the following reasons:
- The electrode slope will change with temperature variations.
- Buffer and sample pH values will change with temperature.
- Measurement drift can occur when the internal elements of the pH and reference electrodes reach thermal equilibrium after a temperature change.
- When the pH electrode and temperature probe are placed into a sample that varies significantly in temperature, the measurements can drift because the temperature response of the pH electrode and temperature probe may not be similar. The sample may not have a uniform temperature, so the pH electrode and temperature probe respond to different environments.
How to Prevent pH Measurement Errors
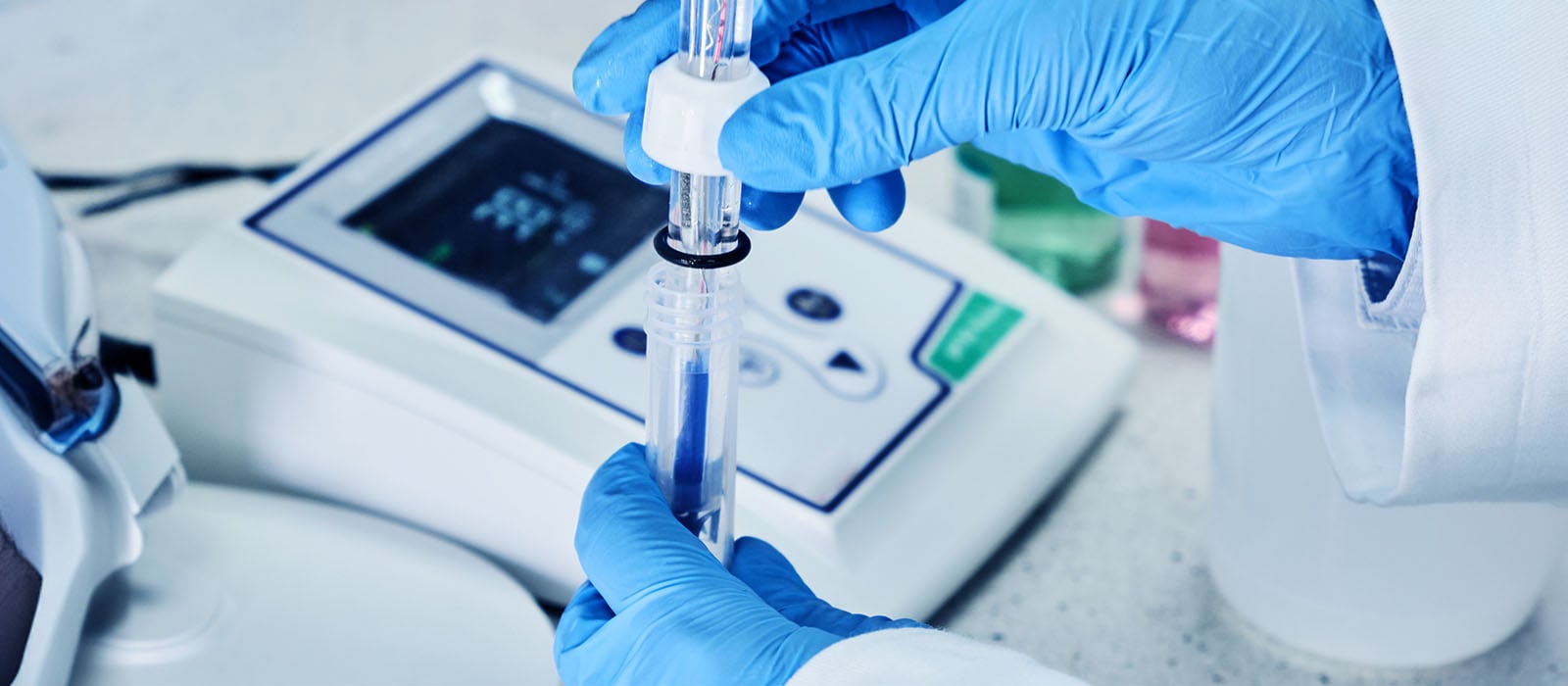
Using an Automatic Temperature Compensation (ATC) Probe
Electrode slope changes can be compensated for by using an ATC probe or a Thermo Scientific Orion Triode electrode with an ATC probe incorporated into the body of the electrode. Thermo Scientific pH meters calculate the electrode slope based on the measured temperature of the pH buffers. The meter will automatically adjust the buffer value to the buffer's actual pH at the measured temperature.
Calibrating the pH Electrode With Buffers
The pH electrode should be calibrated with buffers with known pH values at different temperatures. Since some pH meters without ATC cannot correct sample pH values to a reference temperature, the calibration and measurements should be performed at the same temperature and sample pH values should be recorded with the sample temperature.
This guide will help you understand how pH measurement works and determine the correct steps to avoid making errors when measuring the pH of a solution. Apply everything you’ve learned using the pH measurement instruments available at TLD.
Information provided by Thermo Scientific