Temperature Compensation for pH Instruments
Changes in temperature affect pH. Therefore, when the temperature of a solution changes, it will likely change the solution’s pH. When this happens while measurements are being performed, it does not indicate an error. It simply means that the temperature of the solution has changed.
However, it should be acknowledged that this temperature effect is particularly important in applications where precise pH measurement is critical, such as chemical reactions, biological systems, and industrial processes.
For example, a small change in pH can affect the rate of a chemical reaction or the function of an enzyme. In biological systems, changes in pH can affect the activity of proteins and other biomolecules. In industrial processes, pH can affect the quality of a product, the efficiency of a process, and the safety of workers.
To account for the effect of temperature on pH, it is essential to measure pH at a known temperature and to correct the pH measurement for the temperature of the solution. This correction can be done using a temperature compensation factor that accounts for the change in the dissociation constant of water with temperature.
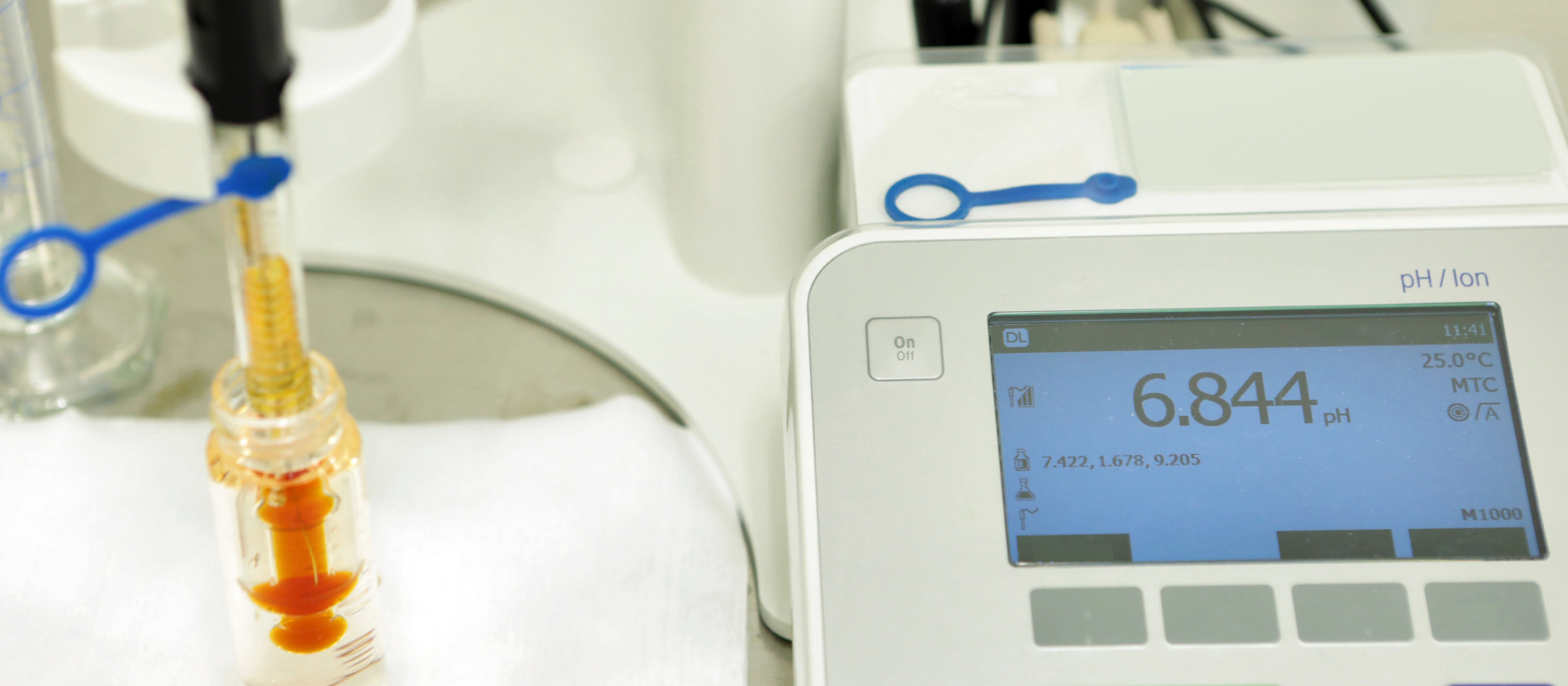
General pH Equipment
Modern pH meters use automatic temperature compensation (ATC) to adjust for the temperature dependency of pH. The pH meter's temperature sensor measures the temperature of the solution being tested, and the meter applies a temperature compensation factor to the pH reading based on the temperature of the solution.
Automatic temperature compensation is essential for accurate pH measurement in many applications, especially when temperature changes are frequent or significant. It allows pH measurements to be made consistently and reliably, regardless of the temperature of the solution being tested.
How It Works
Automatic
This option occurs when a temperature sensor signals to the meter what the solution temperature is, and the meter automatically corrects the pH readings for changes in the electrode sensitivity.
Manual
This option requires the user to dial or key in the solution temperature. The meter then corrects the pH readings for changes in electrode sensitivity.
Temperature Compensation Explained
Temperature compensation eliminates most of the temperature-dependent measurement error potential from an electrode. Further minimization of this error in both temperature-compensating and non-temperature-compensating pH instruments can be achieved by calibrating the instrument and electrode in pH buffers that are close to the expected measurement values for pH and at (or near)the same temperature as the solution to be measured. This technique also minimizes other temperature-related errors that occur in all electrodes but cannot be predicted or compensated for when temperatures change.
These unpredictable pH measurement errors due to temperature changes are somewhat complex in nature and vary from electrode to electrode. They can result in small but noticeable pH measurement errors.
Unfortunately, not much can be done about these kinds of errors in an economical or practical manner for most pH instrument users. Therefore, as a practical matter, they are mostly ignored in everyday pH applications.







