ORP Beginner’s Guide (Oxidation Reduction Potential)

Why Oxidation-Reduction Potential is Important
Oxidation Reduction Potential (ORP) plays a crucial role in water quality testing. ORP is part of an essential group of measurements commonly used to indicate the cleanliness or healthiness of a living body of water. These tests include pH, conductivity, total dissolved solids (TDS), ISP, and salinity.
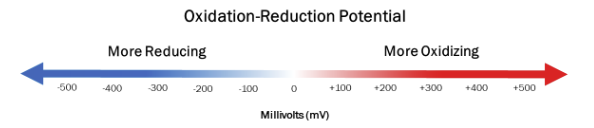
Oxidation Reduction Potential is the potential for electron transfer to occur measured in millivolts (mV) within a medium that causes an oxidation-reduction reaction to occur. ORP measurements with a +mV value correspond to an oxidizing aqueous medium, while a -mV value correlates to a reducing aqueous medium.
For sanitation purposes, an aqueous medium that exhibits a positive ORP is related to a higher level of sanitation that increases as ORP becomes more positive. ORPs that are negative are likely to be increasingly unsanitary and considered unsafe for drinking as the Oxidation Reduction Potential grows more negative. Many oxidants, such as Cl2, NaOCl, ClO2, O3, and UV irradiation techniques, are commonly used to increase ORP to sanitize water, making it safe to drink.
For living bodies of water, such as lakes or streams, the dissolved oxygen (DO) concentration is helpful as an indicator for ORP. DO and other oxidants present in living bodies of water influence Oxidation Reduction Potential making it more positive (oxidizing), subsequently increasing the ability of water to rid itself of waste and other contaminants via oxidation-reduction reactions.
DO is also dependent on temperature, meaning that it also affects ORP. This measurement can also be used to indicate how healthy a living body of water is independently or in conjunction with DO to provide deeper insight.
ORP Meters
ORP meters employ a measuring and a reference electrode that are electrically connected and form a complete circuit when exposed to an electrically conductive solution. An Ag/AgCl reference electrode with a KCl internal solution is commonly used since it produces a constant electrical potential proportional to the KCl internal solution.
When the measuring electrode, typically platinum, is submerged into the sample solution, a voltage potential is generated between it and the reference electrode. The generated voltage potential is then compared to the known voltage potential of the reference electrode in the internal solution to determine the ORP of the sample solution.
ORP meters for evaluating water sanitation and health provide measurements of interest in an industrial or laboratory setting. High-quality, reliable equipment allows you to perform tests with accuracy, precision, and reproducible results. The Lab Depot offers various ORP meters and testing materials to assist your efforts and help you achieve your goals. Explore below all the helpful testing equipment and consumables available at The Lab Depot.
Sources:
Copeland, A.; Lytle, D. A. Measuring the Oxidation-Reduction Potential of Important Oxidants in Drinking Water. Journal - American Water Works Association 2014, 106 (1), E10–E20. https://doi.org/
Suslow, T. V. Oxidation-Reduction Potential (ORP) for Water Disinfection Monitoring, Control, and Documentation; University of California, Agriculture and Natural Resources, 2004. https://doi.org/